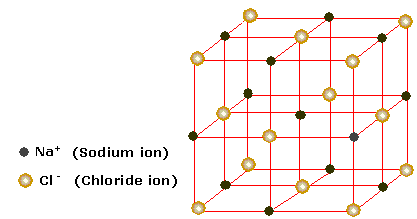
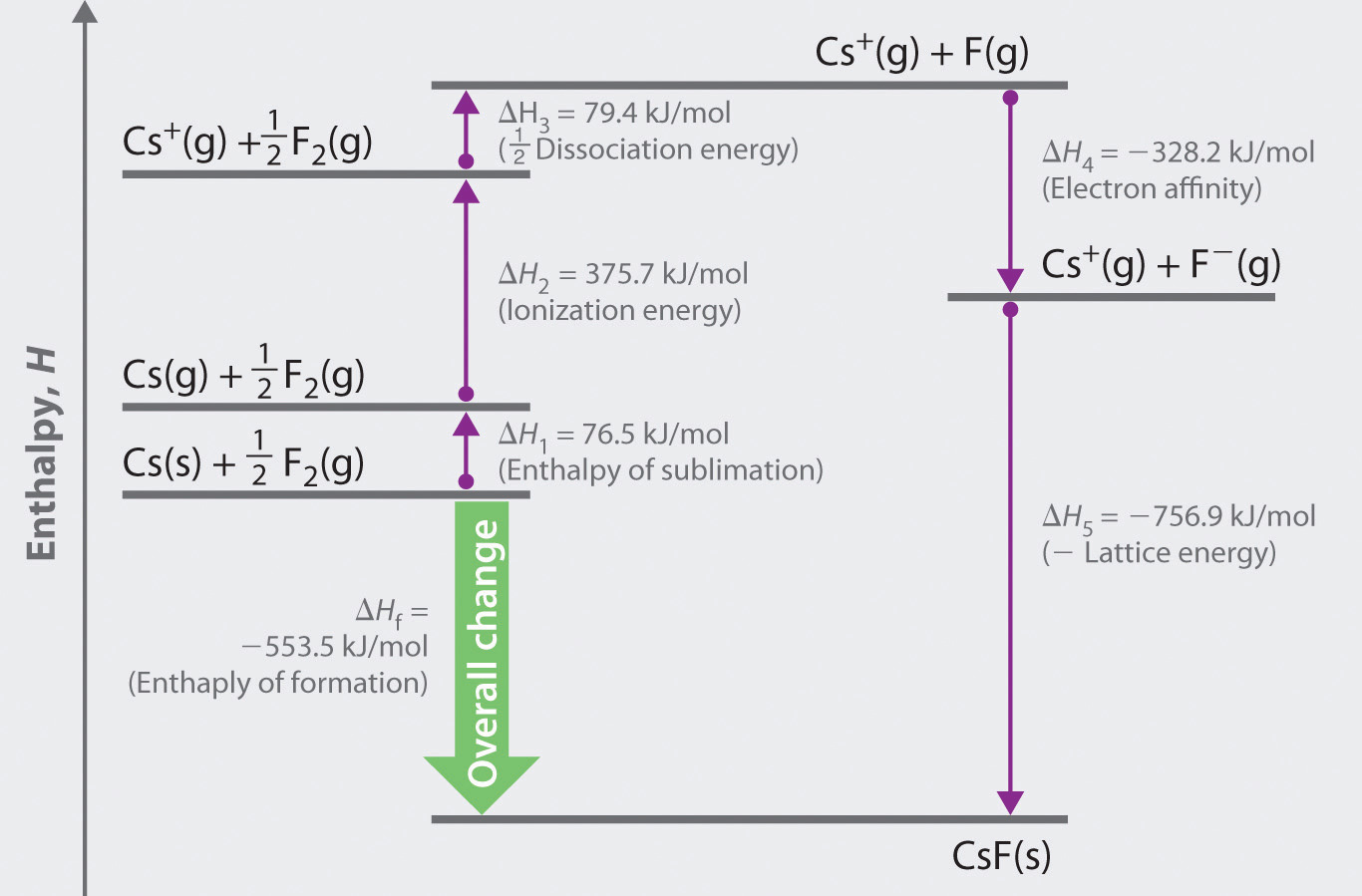
Lattice energy of nacl 89/24/2023 The formation of an ionic compound is exothermic while the dissociation or the reverse of it is endothermic. While the distance between the charges of the ions or the ionic radii increases the lattice energy decreases. The gaseous sodium ion and gaseous chloride ion combine to form a solid sodium chloride molecule and release energy (exothermic) equivalent to the lattice. Hence, lattice energy associated with the crystal AB is 4X.Īs the charges on the ions, that is the cations and the anions, increase, the lattice energy of the compound also increases. 
Therefore, the lattice energy will be 4X. The formula of lattice energy can be derived as $ \right)$ Sodium chloride NaCl is an ionic compound that dissociates as: The ions thus formed are cation (positive ions) and anions (negative ions). For NaCl(s), the lattice energy is defined as the enthalpy of the reaction. Lattice energy can be defined as the energy required to completely separate the ions of an ionic compound into their gas states. An important enthalpy change is the Lattice Energy, which is the energy required to take one mole of a crystalline solid to ions in the gas phase. It is derived by the charges on both ions upon the distance between the two charged atoms in the ionic compound.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
